Description: Which Liquid Makes a Penny Cleaner?
Image copyright: Create-a-Graph.com
The purpose of this lab is to find out what makes a dirty, disgusting penny cleanest. If vinegar has the most acid then it should clean the penny best due to the fact that the acids should eat away at the grime. By putting one penny into these different liquids for a total of seven days we will be able to find out which of the liquids listed will clean a penny better.
Procedures:
1. Take four plastic cups and label them 1, 2, 3, and 4.
2. Take your four plastic cups and fill own with vinegar, one with pop, one with salt water, and one with regular tap water.
3. Observe and record what each penny looks like before you drop it into a cup.
4. Take your dirty pennies and place one into each cup.
5. Make observations on what you see immediately after you drop the pennies into the cups.
6. Let these sit for 7 days and make recorded observations of each penny every day.
7. Make sure to rate each penny on cleanliness for every day you observe and record that information.
Data/Results:
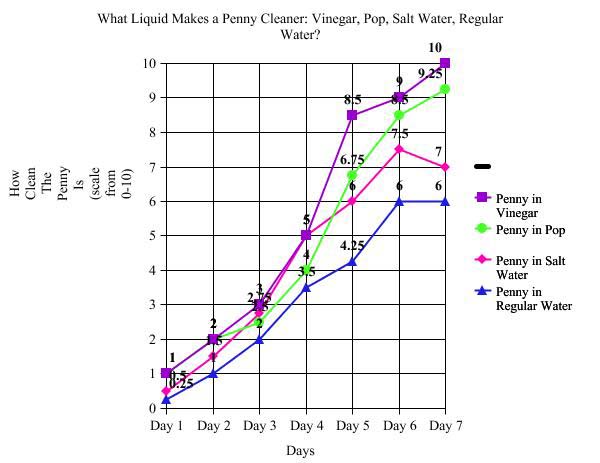
On the first day of my experiment, every penny started at the higher of a 1 on the scale of cleanliness. The second day the penny in pop and vinegar rose to 2 on the scale. The penny in salt water rose to a 1.5 and the penny in the regular tap water went up to a 1. On the third day, vinegar penny is at a 3, followed by the penny in salt water at 2.75, the penny in pop and last the penny in the tap water. The fourth day brought a jump in the vinegar to a 5 along with the salt water penny. Then it was the pop penny with a cleanliness of 4 and the penny in tap is only at 3.5. On the fifth day the vinegar penny was at 8.5, the pop penny at 6.75, the salt water penny at 6, and the tap penny at 4.25. Day six the tap penny was at 6 and the salt water penny was at 7.5. The pop penny was at 8.5 and the vinegar penny was at 9. On the last day of the experiment the vinegar penny was cleaner than all the others with a rating at 10. The salt water penny was at 7, the pop penny was at 9.25, and the tap water was at 6.
Claims and Evidence:
I claim that the vinegar cleaned the penny the best. I know this to be true because throughout the entire week, the vinegar soaked penny was always getting cleaner than the other pennies faster. I also claim that regular tap water worked the worst at cleaning the penny. I know that this is true because during the experimentation week, this penny was the least clean. The final thing I claim is that pop is a good-working supplement for vinegar when it comes to cleaning and making a penny clean. It was second ranked, behind vinegar, during the testing period.
Evidence-Based Explanation:
The data does support the hypothesis. I claim that the vinegar soaked penny is the cleanest after 7 days. I know this is true because after careful testing of one week, the penny went from a miserable 0 out of 10 to 10 out of 10. The reason this occurs is that the acids in vinegar must have eroded away the dirt and grime causing the penny to shine and become clean. Acids are created to eat away at different materials and substances. Therefore if the vinegar has acids in it, which it does, it must have cleaned the penny faster than the others. The other liquids didn\'t have as many acids as the vinegar.
Discussion:
During this experiment I did have some issues and problems with my experiment. The actual reason had nothing to do with the liquids or pennies, but rather with the environment. On the 2nd day of the testing, a family member of mine accidentally knocked the plastic containers over. It did mess up the experiment but mostly wasted 2 very valuable days. I had to restart which took even more time, but in the end I completed the investigation. If I had another chance to redo the experiment, I would place the containers in a remote location where no one could reach or interfere with the testing. Overall I did enjoy the experiment even with the setbacks. It was educational to me and I have learned a lot.